What we offer to patients and their families
We offer a detailed review of the medical history and images by an international team of experts specialized in the care of DIPG/DMGs. This will allow us to provide you with the best current treatment recommendations for your child. We will then schedule a video consultation with you and your family to discuss our recommendations. This could include:
- Biopsy or further resection of the tumors
- Drug screen on patient tumor cells for precision medicine
- Participation in a clinical trial
- Genomic analyses of the tumor tissue

Second opinions
We are offering second opinions after detailed review of medical records, existing molecular data and images. Each patient will be discussed in the trans-Atlantic DIPG/DMG dedicated tumorboard with experts of the University Children’s Hospital Zurich and UCSF. This procedure allows to provide a recommendation for further steps in the medical care based on a broad expert opinion. We can offer second opinions either through in person visits or via telemedicine (video conferencing) in English, French or German, but when needed, we will be happy to set up a phone call with an interpreter.
Please email glioma@kispi.uzh.ch to request a second opinion.

Biopsies for molecular analysis of the tumor


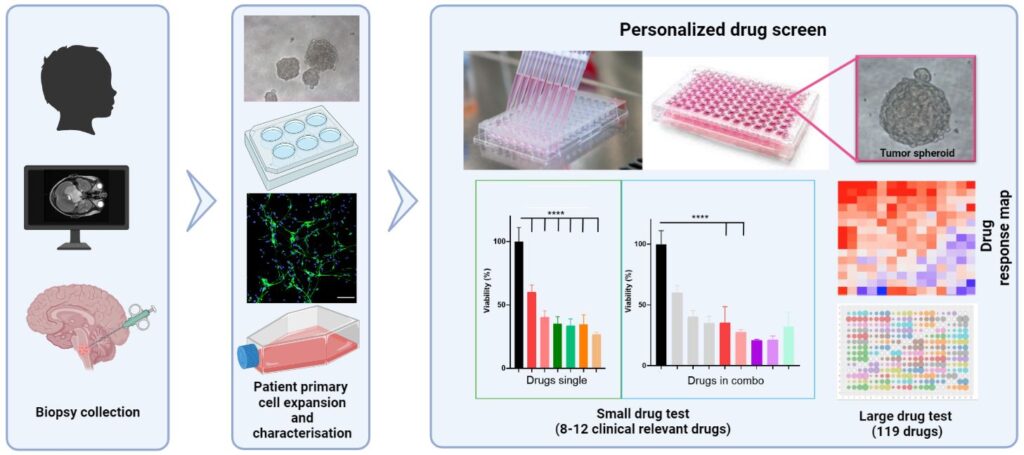
Real time expansion, characterization and drug screen using patient’s derived DMG cells
Drug Screen for precision medicine
Each tumor, like each individual, is unique in its molecular profile. While there are some characteristic patterns in diffuse midline glioma and diffuse intrinsic brainstem glioma, ultimately small differences in cell signaling pathways can make a big difference in terms of drug efficacy. To help us choose the most promising therapeutic approach for your child in the clinic , we can pre-test how tumor cells respond to different drugs in our research laboratory. This process can help to select drugs which fit more precisely to the individual profile.
Clinical trials
At the DIPG/DMG Center Zurich we are actively working on developing and implementing effective and novel clinical trials for patients diagnosed with DIPG and DMG. We are offering clinical trial enrollment but also work with several companies for compassionate use strategies if trial enrollment is not an option.
We are member of the Pacific Pediatric Neuro-Oncology Consortium. Please see all PNOC open trials here: www.pnoc.us

At the DIPG/DMG Center in Zurich we offer enrollment into the following trials.
PNOC022: A Combination Therapy Trial using an Adaptive Platform Design for Children and Young Adults with Diffuse Midline Gliomas (DMGs) including Diffuse Intrinsic Pontine Gliomas (DIPGs) at Initial Diagnosis, Post-Radiation Therapy and at Time of Progression (for patients at the age of 2 – 39)
ClinicalTrials.gov Identifier: NCT05009992
PNOC023: Open label Phase 1 and target validation study of ONC206 in children and young adults with newly diagnosed or recurrent diffuse midline glioma (DMG), and other recurrent malignant brain tumors (for patients at the age of 2 – 21)
ClinicalTrials.gov Identifier: NCT04732065
PNOC019 (currently on hold): A Randomized, Double-Blinded, Pilot Trial of Neoadjuvant Checkpoint Inhibition followed by Combination Adjuvant Checkpoint Inhibition in Children and Young Adults with Recurrent or Progressive High Grade Glioma (HGG)
ClinicalTrials.gov Identifier: NCT04323046
PNOC026: A Phase 2, Open-Label, Multicenter Study to Evaluate the Safety and Efficacy of the Oral Pan-RAF Inhibitor DAY101 in Pediatric Patients with BRAF-Altered, Recurrent or Progressive Low-Grade Glioma
ClinicalTrials.gov Identifier: NCT04775485